
For example, scientists have not been able to resolve what function the HTT gene serves normally, or how its mutation creates problems in the brain. Research on Huntington’s has thus far relied heavily on animal models of the disease, and has left many key questions unanswered. The DNA itself appears in the form of a repeating sequence, and the more repeats there are, the earlier the disease sets in. The mutation takes the form of extra DNA, and causes the gene to produce a longer-than-normal protein. Huntington’s is one of the few diseases with a straightforward genetic culprit: One hundred percent of people with a mutated form of the Huntingtin (HTT) gene develop the disease. The findings have implications for how to best approach treating the disorder, and could ultimately lead to effective therapies. The final domino falls decades after birth, when the symptoms are observable.” “Our research supports the idea that the first domino is pushed soon after fertilization,” Brivanlou says, “and that has consequences down the line. 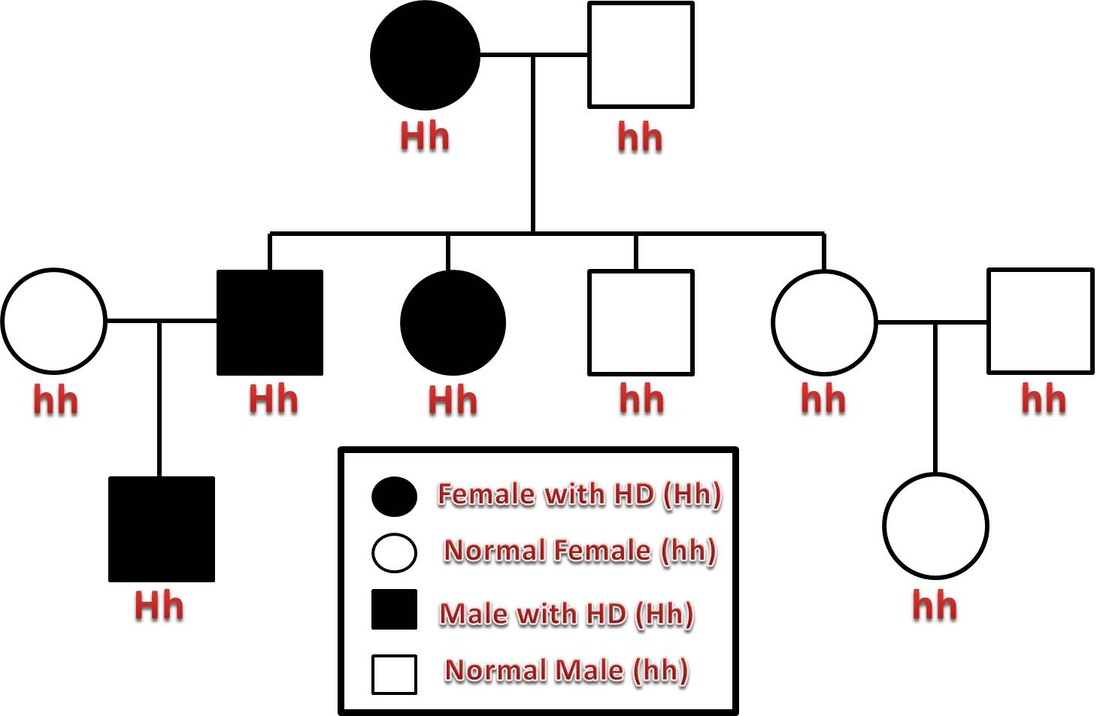
In a report published in Development, they describe early abnormalities in the way Huntington’s neurons look, and how these cells form larger structures that had not previously been associated with the disease. 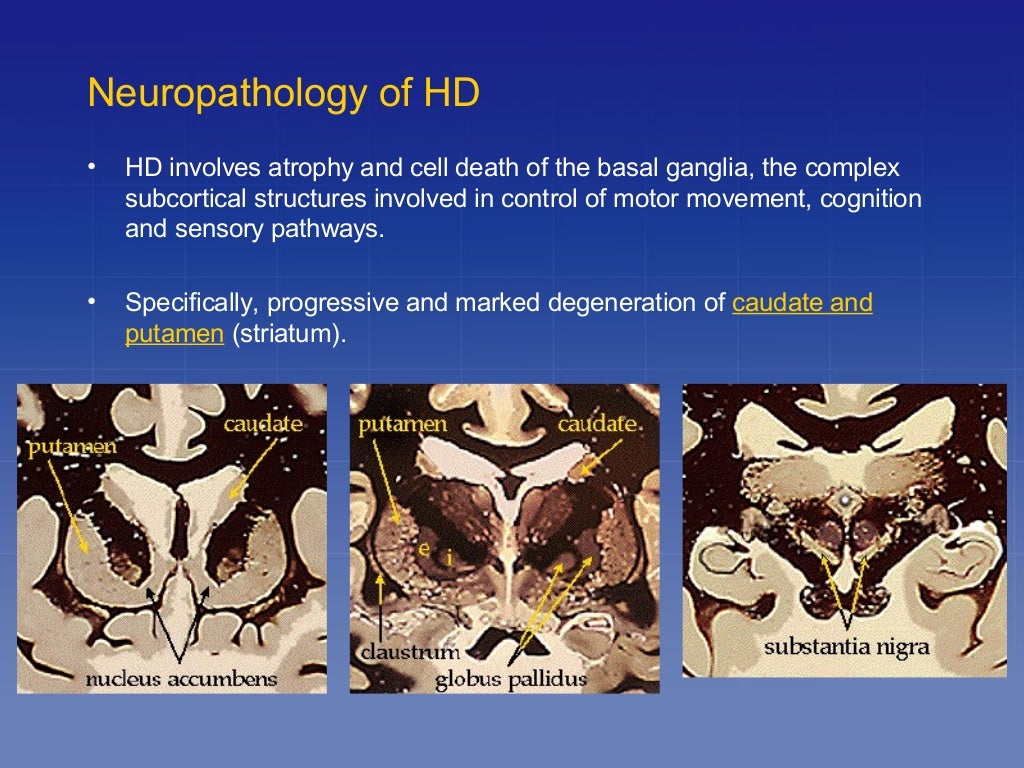
But the new research suggests that these symptoms may be a late manifestation of a disease that originates much earlier, in the first steps of embryonic development.Ī team at Rockefeller led by Ali Brivanlou, the Robert and Harriet Heilbrunn Professor, developed a system to model Huntington’s in human embryonic stem cells for the first time. One in 10,000 Americans suffer from the disease, and most begin to show symptoms in middle age as they develop jerky movements-and as these patients increasingly lose brain neurons, they slide into dementia. With new findings, scientists may be poised to break a long impasse in research on Huntington’s disease, a fatal hereditary disorder for which there is currently no treatment. This may bring us one step closer to treating and potentially preventing this devastating condition.Huntington’s neurons show multiple nuclei (blue) within the same cell, and other signs of trouble, long before symptoms emerge. Antisense oligonucleotide therapy is one such approach with clinical trials currently under way. Perhaps the most promising of these are the emerging therapies aimed at lowering levels of mutant huntingtin. In the last decade there has been a large growth in potential therapeutic targets and clinical trials. In recent years there have been significant advances in understanding both the cellular pathology and the macroscopic structural brain changes that occur as the disease progresses. There are currently no disease modifying treatments therefore supportive and symptomatic management is the mainstay of treatment. Early macroscopic changes are seen in the striatum with involvement of the cortex as the disease progresses. At the cellular level mutant huntingtin results in neuronal dysfunction and death through a number of mechanisms, including disruption of proteostasis, transcription and mitochondrial function and direct toxicity of the mutant protein. It is characterized by cognitive, motor and psychiatric disturbance. In Western populations HD has a prevalence of 10.6–13.7 individuals per 100 000. Huntington's disease (HD) is a fully penetrant neurodegenerative disease caused by a dominantly inherited CAG trinucleotide repeat expansion in the huntingtin gene on chromosome 4.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
